|
4/16/2024 0 Comments Density of water gcmThe Roman numerals indicate various ice phases. Freezing water expands over 9 by volume and ice floats on water because it is lighter. At 4 degrees Celsius pure water has a density of 1g/mL or 1kg/L and a specific gravity of 1. The density of water varies according to temperature and the degree of purity. ‡ Ice XI triple point is theoretical and has never been obtained Phase diagram Log-lin pressure–temperature phase diagram of water. Density of pure water is a constant at a certain temperature not depending on sample. Table of various forms of ice Properties of various forms of ice Melting point of ice at various pressures ĭata obtained from CRC Handbook of Chemistry and Physics 44th ed., p. 2390 The sixth column is the density of the vapor. The fifth column is the work PΔ V done by each gram of liquid that changes to vapor. The fourth column is the heat of vaporization of each gram of liquid that changes to vapor. The third column is the heat content of each gram of the liquid phase relative to water at 0 ☌. Pressure of the equilibrium is given in the second column in k Pa. Where P is equilibrium vapor pressure in k Pa, and T is temperature in kelvins.įor T = 273 K to 333 K: A = 7.2326 B = 1750.286 C = 38.1.įor T = 333 K to 423 K: A = 7.0917 B = 1668.21 C = 45.1.ĭata in the table above is given for water–steam equilibria at various temperatures over the entire temperature range at which liquid water can exist. What is the SI unit of relative density (c) The density of turpentine is 840 kg/m 3. Vapor pressure formula for steam in equilibrium with liquid water: log 10 P = A − B T − C, What is the SI unit of density (b) Define relative density. The values below 0 ☌ refer to supercooled water.Įlectrical conductivity of highly purified water at saturation pressure Temperature, ☌ Liquid physical properties Temperature dependence of the surface tension of pure water Temperature dependence of the density of ice and water Velocity of sound in water  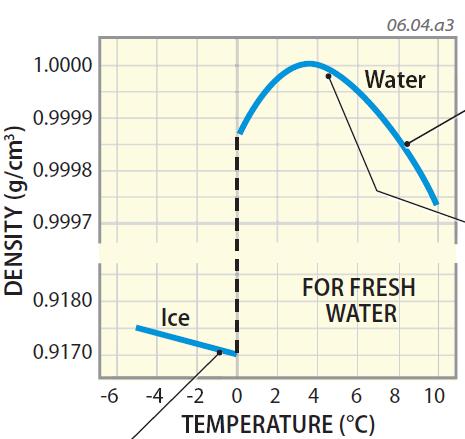
At this temperature, a gallon of water weighs around 8.345 lbs. Definitions, online calculator and figures and tables with water properties like density, specific weight and thermal expansion coefficient of liquid water at temperatures ranging 0 to 360C (32 to 680F). Water has a maximum density of 39.2✯ or 4✬. Water - Density, Specific Weight and Thermal Expansion Coefficients. This is equal to a rounded value of 1 gram per milliliter (g/ml) or 1 gram per cubic centimeter (g/cm 3) or 1000 kg/m 3. K) at 80 ☌ħ5.74 J/(mol K) and 4.2050 J/(g The density of water at 4 degrees Celsius is 8.345 lbs/U.S.Water is the only substance where the maximum density does not occur when solidified. Fresh water has a maximum density at around 4° Celsius. When water freezes it expands rapidly adding about 9 by volume. K) at 50 ☌ħ5.37 J/(mol K) and 4.1843 J/(g Water is the only substance on Earth that exists in all three physical states of matter: solid, liquid and gas.Thermodynamic properties Phase behaviorħ5.97 J/(mol K) and 4.2176 J/(g Structure and properties Structure and propertiesĤ92.215 kJ/mol O–H bond dissociation energy By the time the surface water reaches 4 ☌ (39. This indicates the units that must be divided into the quantity to yield the pure number in the table or on the axis.This page provides supplementary data to the article properties of water.įurther comprehensive authoritative data can be found at the NIST Webbook page on thermophysical properties of fluids. During the fall a lake is cooled at its surface, the surface water sinks, and convective overturn proceeds as the density of the surface water increases with the decreasing temperature. Understanding the density of water is important in various fields, including engineering, chemistry, biology, and. It is typically measured using techniques such as hydrometry, hydrostatic weighing, and Archimedes’ principle. For example, liquid water has a density of 1.00 g/mL (or 1. The density of water in g/cm3 is a fundamental physical property that changes with temperature and pressure. Therefore, a column in a table or the axis of a graph is conveniently labeled in the following form: Density is a property of the material itself, so each unique substance has its own corresponding density, which can vary slightly with temperature. \): Density of Several Substances at 20☌.Anchor Substance
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
